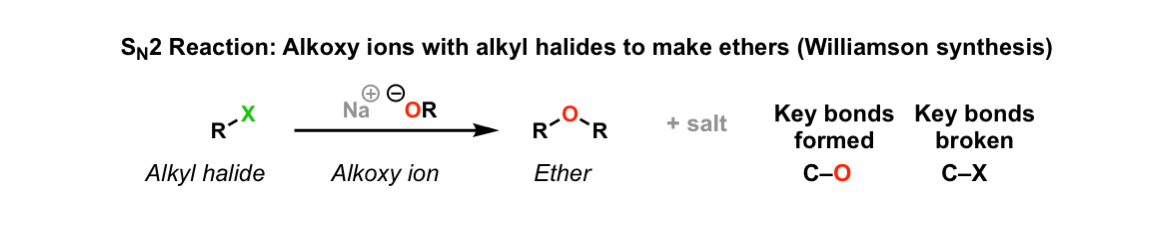
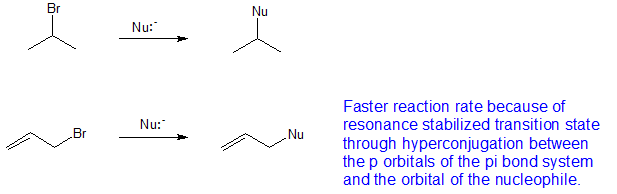
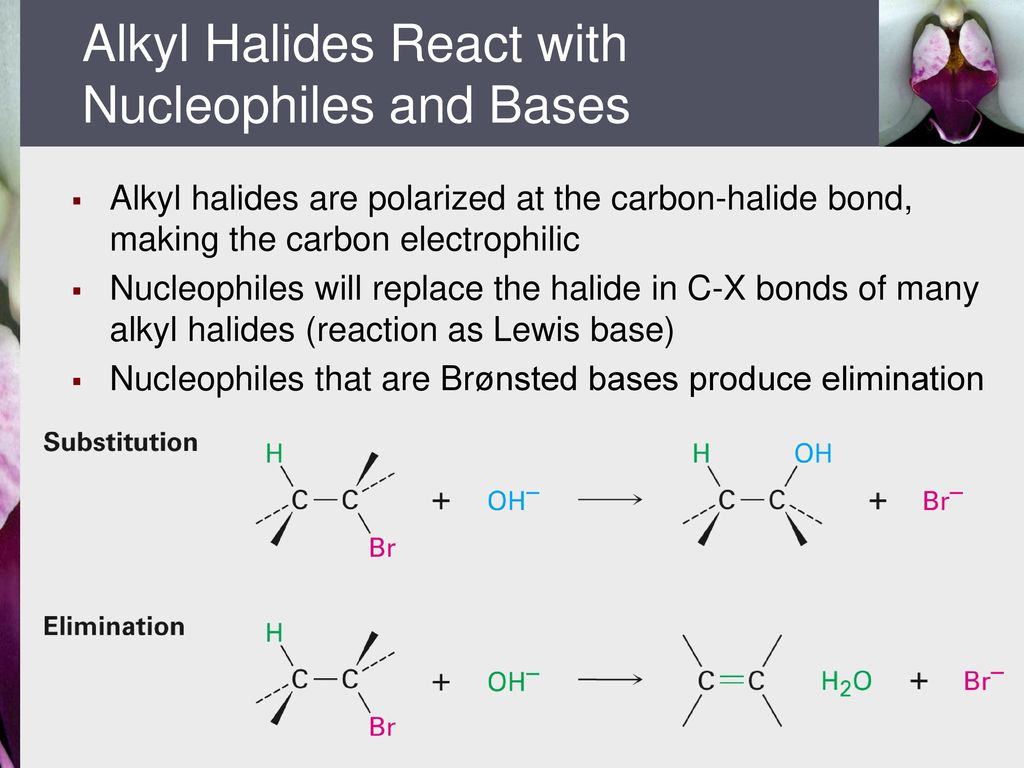
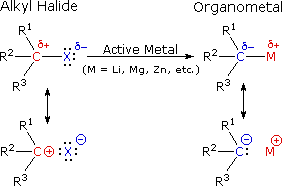Such conformers are therefore present in extremely low concentration and the rate of elimination is very slow.
Why alkyl halides are more reactive than vinyl halides.
For s n 1.
This is because c x bond in vinyl halides have partial double bond character due to resonance.
Vinyl halides are less reactive than alkyl halides.
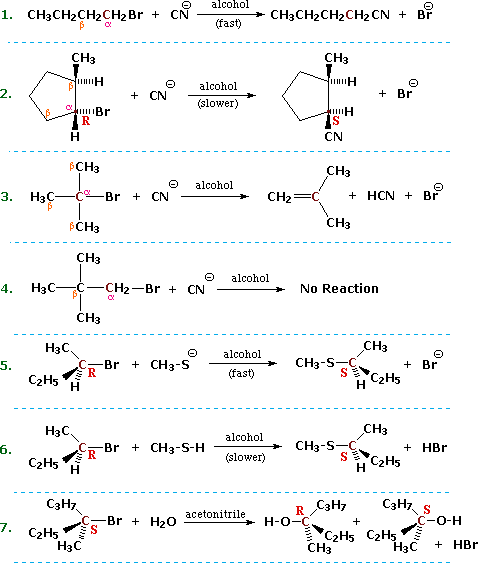
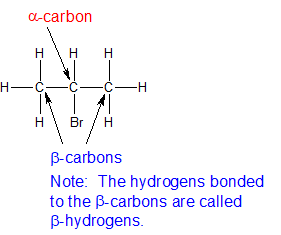
It is assumed that the alkyl halides have one or more beta hydrogens making elimination possible.
And that low dielectric solvents e g.
This causes shortening of bond length in aryl.
Halogen elements definition properties reactivity and uses.
The sp2 hybridised carbon atom with greater s character is more electronegative.
It can hold the electron pair of the bond more tightly than sp3 hybridised carbon atom in alkyl halides with less s character.
Because of this polarization carbon becomes electron deficient and alkyl halides become more reactive.
When a high dielectric solvent would significantly influence the reaction this is noted in red.
C aryl halides are less reactive than alkyl halide d aryl halides are more reactive than benzyl halides note.
Alkyl halides are more reactive due to the polarised bond of r x.
Follow report by harshtomar9049 06 12 2019 log in to add a comment.
It takes very lots.
Learn in our free webinar.
In the following questions a s tatement of assertion followed by a statement of reason is given.
The aryl halides have the bonded electrons in a decrease skill sp2 hybrid orbital greater s character decrease skill than all the different varieties that are all in sp3 hybrid orbitals.
This is because c x bond in vinyl halides have par how to create a game using turtle python library.
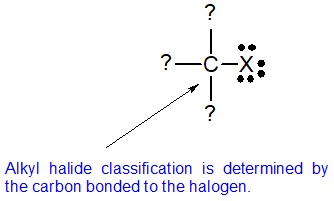
In alkyl halides carbon atom of c x bond is sp3 hybridised while in aryl halides the carbon atom attached to halogen is sp2 hybridised.
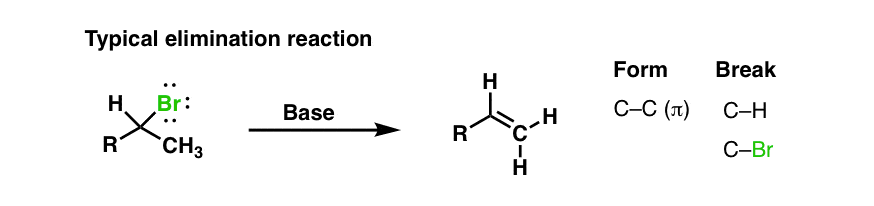
Having arrived at a useful and plausible model of the e2 transition state we can understand why a bulky base.
Tertiary carbocations are greater stable as a results of hyperconjugating impacts of adjacent alkyl communities it is likewise why secondary halides are greater reactive than typical.